Gene Bio Systems
Long PCR Taq DNA polymerase
Long PCR Taq DNA polymerase
SKU:P1061
Couldn't load pickup availability
Long PCR Taq DNA Polymerase,a combination of two thermostable DNA polymerases, Taq and Pfu, is a special formulation designed for amplifying large fragment. This specially formulated Long PCR Taq was shown to amplify long templates from λ phage genome of up to 20 kb. It is also a better choice for amplifying complex template, such as GC-rich template.
Long PCR Taq is suitable as a direct replacement for ordinary Taq Polymerase in most applications. Using Long PCR Taq in your PCR reactions results in 3´-dA overhangs PCR products, which can be used in TA clone
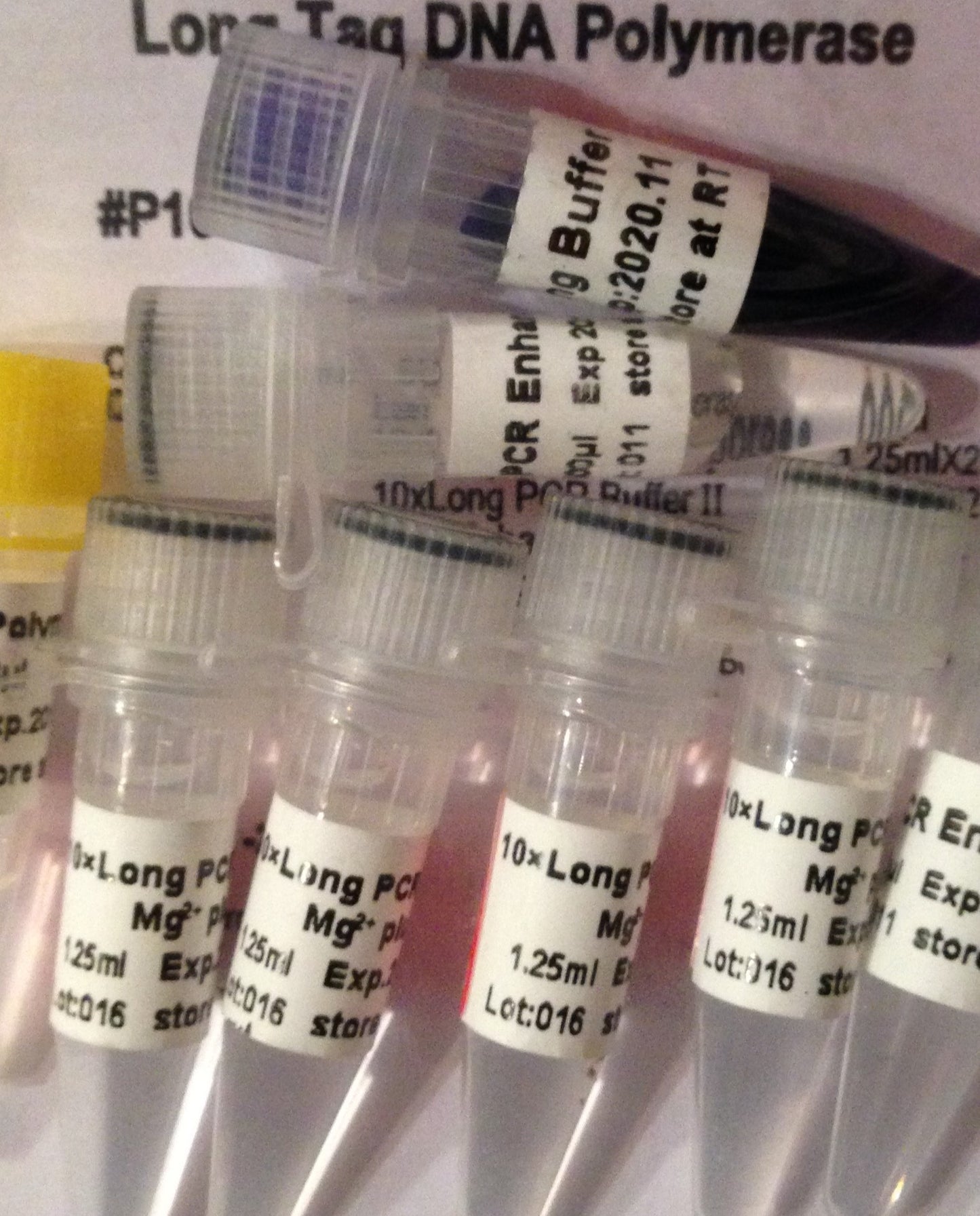
Contents: Long PCR Taq DNA Polymerase, PCR Enhancer, 6x gel loading buffer, 10X Long PCR Taq Buffer Ⅰ with Mg2+, 10X Long PCR Taq Buffer Ⅱ with Mg2+
Applications
• PCR amplification of DNA fragments any sizes around 5 kb
• DNA labeling
• DNA sequencing
• PCR for cloning
Unit Definition
One unit is defined as the amount of the enzyme required to catalyze the incorporation of 10 nmoles of dNTP’s into an acid-insoluble form in 30 minutes at 70°C using hering sperm DNA as substrate.
Storage Buffer
20mM TrisCl ( pH8.0), 100mM KCl, 3mM MgCl2 1mM DTT,0.1% NP-40, 0.1% Tween20, 0.2mg/ml BSA, 50% (v/v) glycerol
10X Long PCR Taq Buffer Ⅰ with Mg2+
500mM Tris-HCl pH 8.8,160mM (NH4)2SO4 ,25mM MgCl2 ,1% Triton X-100
10X Long PCR Taq Buffer Ⅱ with Mg2+
200mM Tris-HCl PH8.8,100mM KCl,100mM (NH4)2SO4,16mM MgSO4,1% Tritonx-100
Features
• High fidelity: three times fidelity of Taq DNA Polymerase.
• Longer fragment: amplify long templates as long as 40kb.
• Amplification of complex template (GC rich or repetitive sequence).
• Generates 3'-dA and blunt end PCR products.
Note:
- 10xLong PCR BufferⅠis classical Long PCR Taq DNA Polymerase buffer, is good for long template especially above 10kb.
- 10xLong PCR Buffer Ⅱ is an alternatie long PCR buffer . It is for better fidelity but may not be robust for longer templates above 10kb.
• Users may choose compare the two buffers for different template.
Basic PCR Protocol
The following basic protocol serves as a general guideline and a starting point for any PCR amplification. Optimal reaction conditions
(incubation time and temperature, concentration of Taq DNA Polymerase, primers, Mg2+, and template DNA) vary and need to be optimized.
1. Add the following components to a sterile microcentrifuge tube sitting on ice:
|
Reagent |
Volume (50 µl rxn) |
Final concentration |
|
10x PCR Buffer |
5 µl |
1x |
|
dNTPs(10 mM each) |
1 µl |
0.2 mM each |
|
Primer I |
Variable |
0.4-1 µM |
|
Primer II |
Variable |
0.4-1 µM |
|
Long PCR DNA polymerase (5U/µl) |
0.25-0.5 µl |
1.25-2.5U/50 µl |
|
Water |
Variable to 50 µl |
N.A. |
2. Mix contents of tube. Cap tubes and centrifuge briefly to collect the contents to the bottom. When using a thermal cycler that does not
contain a heated lid, overlay the reaction mixture with 25 μl mineral oil.
3. Perform 25-35 cycles of PCR amplification as follows:
|
Initial Denaturation |
94°C |
3 min |
|
25-35 Cycles |
94°C 55-68°C 72°C |
30s 30s 1-10 mins |
|
Final Extension |
72°C |
10 min |
4. Incubate for an additional 10 min at 72°C and maintain the reaction at 4°C. The samples can be stored at -20°C until use.
5. Analyze the amplification products by agarose gel electrophoresis and visualize by ethidium bromide staining. Use appropriate molecular weigh standards.
Notes on cycling conditions
- Initial denaturation can be performed over an interval of 1~5 min at 95℃ depending on the GC content of template.
-Denaturation for 30 sec to 2 min at 94~95℃ is sufficient. If the amplified DNA has a very high GC content, denaturation time may be increased up to 4 min.
-Optimal annealing temperature is 5℃ lower than the melting temperature of primer-temperature DNA duplex. If nonspecific PCR products are obtained optimization of annealing temperature can be performed by increasing temperature stepwise by 2℃.
-The number of PCR cycles depends on the amount of emplate DNA in the reaction mix and on the expected yield of the PCR products, 25-35 cycles are usually sufficient
for the majority PCR reaction. Low amounts of starting template may require 40 cycles.
-The time of the final extensi, on step can be extended for amplicons that will be cloned into T/A vectors.